Bioenergetic Regulation: Advancing Research in Lipolysis, ATP Synthesis, and Metabolic Flux
30th Mar 2026
This comprehensive guide explores a specialised selection of peptides and small molecules curated by Amino Peptides Ltd, designed specifically to investigate the complexities of energy homeostasis—the physiological master-switch governing energy intake, storage, and expenditure.
In the modern research landscape, understanding the "metabolic phenotype" is paramount. Researchers and clinical investigators utilise these reagents to dissect the fundamental mechanisms of cellular life, from the rapid mobilisation of stored adipose tissue (lipolysis) to the intricate dance of the Electron Transport Chain (ETC) within the mitochondria. These compounds are indispensable for studies exploring the global rise of obesity, metabolic syndrome, and the inevitable decline of mitochondrial efficiency associated with biological ageing.
The Dual Pillars of Metabolic Research: Adiposity and Bioenergetics
Metabolic research is generally categorised into two primary vectors: Adipose Tissue Modulation (the breakdown and oxidation of stored lipids) and Mitochondrial Optimisation (the generation and conservation of cellular energy). While these fields were once studied in isolation, we now understand them as a singular, integrated system where mitochondrial health dictates the efficiency of fat oxidation.
1. Lipolysis and the Mobilisation of Adipose Tissue
The study of obesity often focuses on the "lipolytic" pathway—the biochemical process by which triglycerides are hydrolysed into glycerol and free fatty acids.
The AOD-9604 and HGH Fragment 176-191 Pathway
Among the most studied reagents in this category are AOD-9604 and HGH Fragment 176-191. These compounds represent a surgical approach to endocrinology. By isolating the C-terminus of the human growth hormone molecule (tyrosine at the 176th position to phenylalanine at the 191st), researchers can study the potent fat-burning properties of HGH without the confounding variables of insulin-like growth factor (IGF-1) elevation or decreased insulin sensitivity.
Research indicates that these peptides interact with beta-adrenergic receptors on adipocytes, stimulating the activity of Hormone-Sensitive Lipase (HSL). This enzyme is the gatekeeper of fat storage; once triggered, it breaks down the triglycerides stored in white adipose tissue (WAT). This mechanism is central to current research into "targeted" fat loss, specifically seeking to resolve the stubborn metabolic resistance found in visceral fat depots.
Tesamorelin: Targeting Visceral Adiposity
While subcutaneous fat is often a cosmetic concern, Tesamorelin remains the gold standard for investigating Visceral Adipose Tissue (VAT). Visceral fat is metabolically active in the worst way, secreting pro-inflammatory cytokines that drive insulin resistance. Tesamorelin, a Growth Hormone Releasing Hormone (GHRH) analogue, is researched for its unique ability to selectively reduce this deep-seated tissue, thereby improving triglyceride profiles and systemic metabolic health in research models.
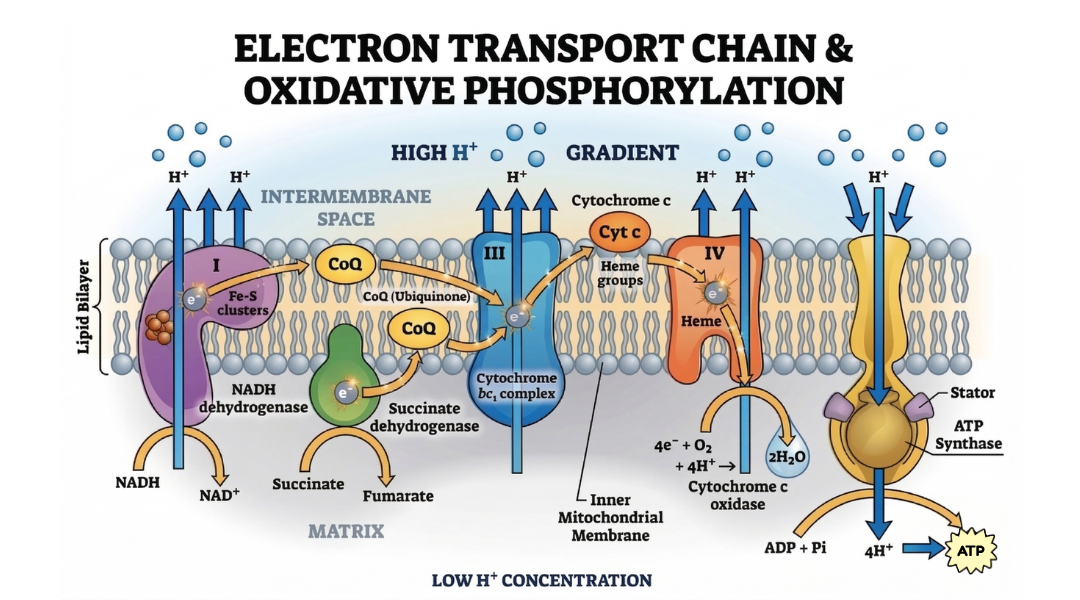
2. Mitochondrial Biogenesis: The MOTS-c Revolution
The mitochondria are no longer viewed simply as "powerhouses" but as signalling hubs that dictate the fate of the cell.
MOTS-c: The Mitochondrial-Derived Peptide (MDP)
One of the most exciting developments in bioenergetics is the discovery of MOTS-c (Mitochondrial Open Reading Frame of the 12S rRNA-c). Unlike traditional hormones, MOTS-c is encoded within the mitochondrial genome itself. It acts as a metabolic "messenger," translocating to the nucleus in response to metabolic stress to regulate genes involved in glucose handling.
MOTS-c is a potent activator of AMPK (Adenosine Monophosphate-activated Protein Kinase). Known as the body’s "metabolic master switch," AMPK activation mimics the physiological effects of high-intensity exercise. In research settings, MOTS-c has been shown to:
-
Enhance glucose uptake in skeletal muscle.
-
Promote fatty acid oxidation.
-
Reverse diet-induced insulin resistance.
3. Cellular Energy Flux and NNMT Inhibition
As we delve deeper into the molecular level, the focus shifts to the cofactors that allow these metabolic processes to occur: NAD+ and the enzymes that regulate its availability.
NAD+: The Universal Electron Carrier
Nicotinamide Adenine Dinucleotide (NAD+) is the essential substrate for ATP production and the primary fuel for Sirtuins—the family of enzymes responsible for DNA repair and longevity. Research into metabolic decline often reveals a "NAD+ sink," where the body consumes more NAD+ than it can produce, leading to mitochondrial "misfiring" and cellular senescence.
5-Amino-1MQ: The NNMT Inhibitor
A significant barrier to maintaining high NAD+ levels is an enzyme called Nicotinamide N-methyltransferase (NNMT). NNMT is highly expressed in the white adipose tissue of individuals with obesity and diabetes, where it acts as a "metabolic brake," slowing down the rate at which cells burn energy.
5-Amino-1MQ is a small molecule designed to inhibit NNMT. By blocking this enzyme, researchers observe a dramatic increase in intracellular NAD+ levels and a concomitant decrease in the growth of white adipose tissue. This represents a "bottom-up" approach to metabolic efficiency: by removing the enzymatic brakes, the cell's natural capacity for thermogenesis is restored.
4. Synergistic Research Protocols: SS-31 and Beyond
In advanced metabolic studies, researchers often combine reagents to investigate synergistic effects on cellular "resurrection."
The Mitochondrial Restoration Protocol (SS-31 + NAD+)
The combination of SS-31 (Elamipretide) and NAD+ is currently the gold standard for investigating the reversal of age-related mitochondrial decline. To use an engineering analogy: if NAD+ is the fuel, SS-31 is the mechanic that repairs the engine.
ATP Production x Mitochondrial Membrane Potential (Δ╰|╯?)
SS-31 works by specifically targeting and stabilising Cardiolipin, a unique phospholipid found in the inner mitochondrial membrane. Cardiolipin is essential for the structural integrity of the cristae; when it oxidises, the mitochondria leak electrons, leading to oxidative stress and reduced ATP output. By pairing SS-31 with NAD+, researchers can investigate whether restoring mitochondrial structure allows for more efficient energy production in senescent cell models.
5. Quality Assurance in Metabolic Science
At Amino Peptides Ltd, we recognise that the validity of metabolic data is entirely dependent on the purity of the reagents used. Metabolic pathways are incredibly sensitive to external stimuli; even trace amounts of bacterial endotoxins can trigger an immune response that masks the true metabolic effect of a peptide.
Our commitment to quality is underpinned by a rigorous three-tier testing protocol:
-
High-Performance Liquid Chromatography (HPLC): We guarantee a purity level of >99%, ensuring that dose-response curves in your research are accurate and reproducible.
-
Liquid Chromatography-Mass Spectrometry (LC-MS): This confirms the exact molecular mass and identity of the peptide, ensuring no structural variations exist.
-
Endotoxin Control: Critical for mitochondrial assays where bacterial byproducts could skew inflammatory markers or oxygen consumption rates (OCR).
-
Lyophilisation Stability: Peptides like MOTS-c and NAD+ are highly susceptible to hydrolytic degradation. Our reagents are vacuum-sealed in a lyophilised (freeze-dried) state to ensure maximum shelf-life and potency upon reconstitution.
Conclusion: The Future of Energy Homeostasis
The transition from treating metabolic symptoms to addressing the underlying bioenergetic causes is the current frontier of science. Whether your research focuses on the lipolytic potential of AOD-9604, the mitochondrial signalling of MOTS-c, or the enzymatic inhibition of 5-Amino-1MQ, the tools provided by Amino Peptides Ltd offer the precision required for world-class discovery.
Technical References and Primary Literature
-
M Heffernan 1, R J Summers, A Thorburn, E Ogru, R Gianello, W J Jiang, F M Ng. "The effects of human GH and its lipolytic fragment (AOD9604) on lipid metabolism following chronic treatment in obese mice and beta(3)-AR knock-out mice. " National Library of Medicine. View Study
-
Lee, C., et al. (2015). "The Mitochondrial-Derived Peptide MOTS-c Promotes Metabolic Homeostasis and Reduces Diet-Induced Obesity." Cell Metabolism. View Study
-
Note: The definitive study on MOTS-c and its role in AMPK activation and glucose regulation.
-
Kraus, D., et al. (2014). "Nicotinamide N-methyltransferase promotes excessive accumulation of fat and is a viable target for treating obesity." Nature Communications. View Study
-
Note: This research validates the inhibition of NNMT (the pathway used by 5-Amino-1MQ) as a method for increasing NAD+ and reducing adiposity.
-
Muhammad Shahzeb Khan, Javed Butler (2019). "Elamipretide Improves Mitochondrial Function in the Failing Human Heart" Journal of the American College of Cardiology. View Study
